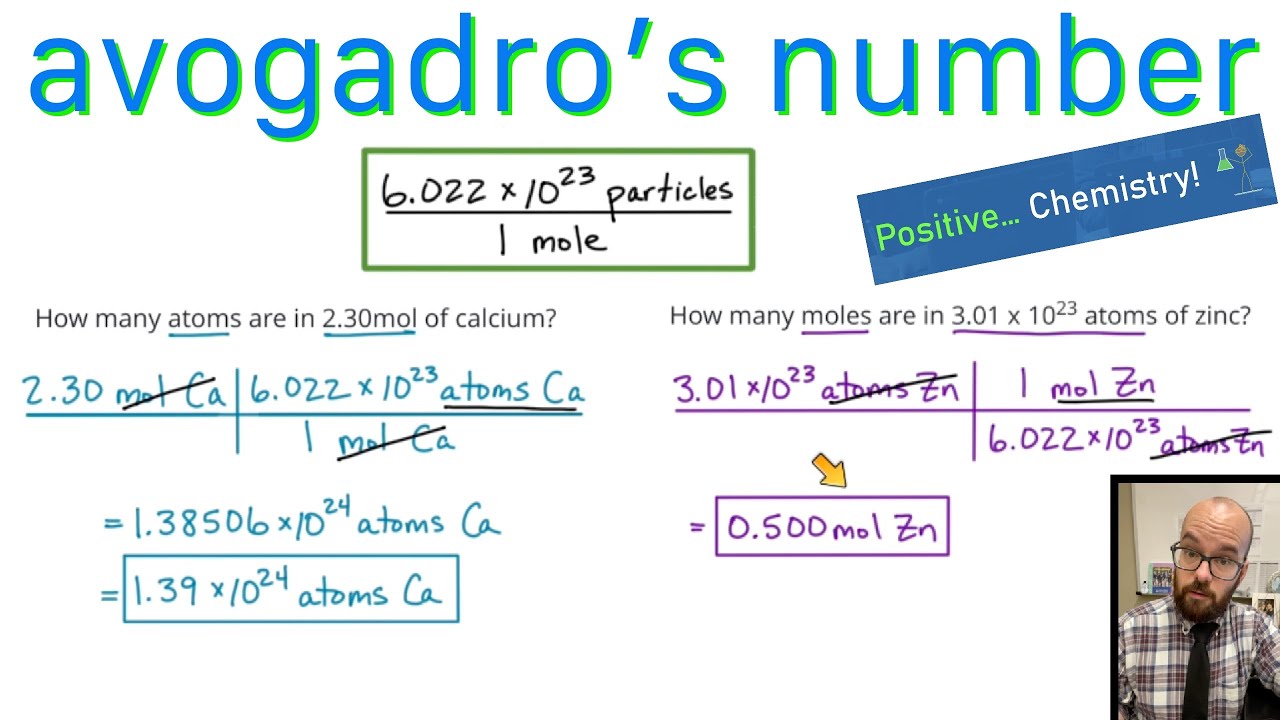
1. Using Avogadro's number, calculate the number of atoms in 0.005 kilograms of carbon. 2. If there are 'x' atoms in 5 grams of carbon, how many atoms are there in 5
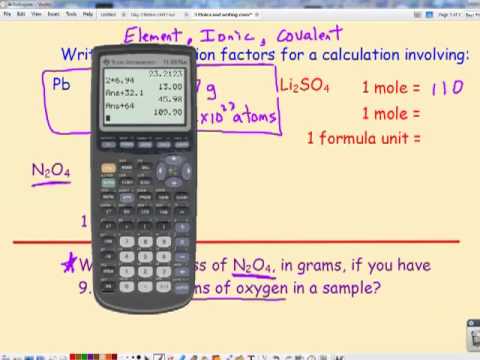
Is there a preset function in Texas Instrument-84 Plus calculators for Avogadro's number? Can you add one? - Quora

1. Using Avogadro's number, calculate the number of atoms in 0.005 kilograms of carbon. 2. If there are 'x' atoms in 5 grams of carbon, how many atoms are there in 5

1. Using Avogadro's number, calculate the number of atoms in 0.005 kilograms of carbon. 2. If there are 'x' atoms in 5 grams of carbon, how many atoms are there in 5

Use Avogadro's Number to Calculate the Mass of a Single Atom | Relative atomic mass, Chemistry, Atom

Physical Chemistry #1: Relative Mass, the Mole and Avogadro's Constant (Slides & Student Led Tasks) | Teaching Resources

Converting Between Particles and Moles Using Avogadro's Number and Conversion Factors – Science and Joe